Novel Antiviral Approach To Covid-19 Treatment
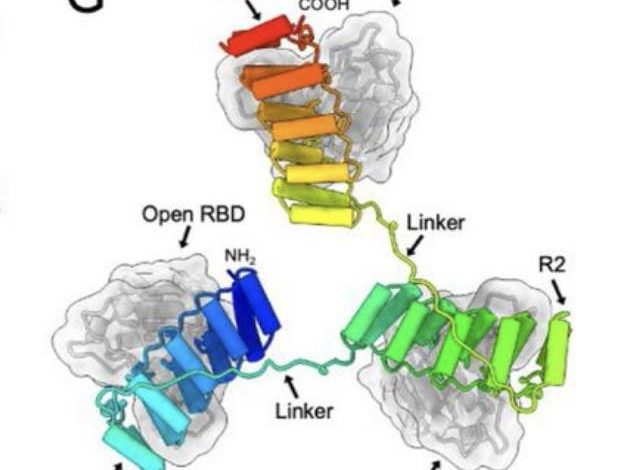
Mannequin of the three RBD-targeting DARPin molecules of ensovibep sure to the RBD areas of the Spike … [+]
FROM: “ENSOVIBEP, A NOVEL TRISPECIFIC DARPIN CANDIDATE THAT PROTECTS AGAINST SARS-COV-2 VARIANTS” SYLVIA ROTHENBERGER ET AL. 2021.
As we transfer into the third yr of the Covid-19 pandemic, issues proceed to look pretty bleak. Warning fatigue mixed with the concept that omicron is “delicate” have left us with document case numbers and record hospitalizations. However on the remedy entrance, we’re nonetheless lagging, with omicron and different variants proof against a lot of our at the moment out there antiviral therapies. That mentioned, Swiss pharmaceutical company Novartis, working along with biotech group Molecular Companions, has simply thrown its hat within the ring, and early trial results for his or her new drug, ensovibep, appear promising.
Coming off the again of a profitable phase 1 safety study in March of 2021, Novartis and Molecular Companions initiated a world medical trial referred to as EMPATHY. This part 2 and three examine goals to check the security and efficacy of ensovibep in symptomatic Covid-19 sufferers in a non-hospitalized setting. Ensovibep is run as a single-dose intravenous infusion.
The newest outcomes come from part 2, which centered on preliminary efficacy and on figuring out the optimum dose. To this finish, 407 grownup sufferers have been recruited and randomized into 4 completely different arms: one for every dose examined, and one placebo management group. All sufferers displayed no less than two signs of delicate to average Covid-19 —fever, cough, physique aches, shortness of breath, and so on.— and represented a mixture of each vaccinated and unvaccinated people. Their Covid-19 standing was confirmed by way of constructive fast antigen take a look at the day of dosing in addition to an extra PCR take a look at at baseline. Dosing befell inside seven days of preliminary symptom onset.
The trial met its main endpoint, displaying a marked discount in viral load throughout the span of eight days. It additionally met the secondary endpoint, with a 78% discount in hospitalizations, visits to the emergency room, or dying when in comparison with the placebo group. This held true throughout the entire doses examined: 75mg, 225mg and 600mg. Importantly, all three doses have been described as being well-tolerated by the sufferers, providing an excellent security profile. Novartis plans on shifting ahead with the 75mg dose.
After the outcomes of the primary trial part got here by means of in Might of 2021, Patrick Amstutz, Chief Govt Officer of Molecular Companions, said: “By advantage of its tri-specific design, ensovibep was constructed to withstand viral mutations and certainly exhibits potent inhibition of all variants of concern up to now, with the potential to keep up exercise additionally for future variants.”
Mechanism of Motion
Ensovibep is based on designed ankyrin repeat proteins (DARPins): genetically engineered proteins that mimic an antibody’s means to bind to, and ultimately neutralize, antigens. DARPins are derived from pure ankyrin repeat proteins, a category of binding protein very steadily present in nature. They are often designed round their precise goal and their supposed impact, permitting for a substantial amount of specificity. This ends in a high-strength, tight-fitting bond with their goal molecule.
Within the case of ensovibep, there are 5 completely different DARPin domains linked collectively as one single molecule (FIGURE 1A); two of those bind to human serum albumin (HSA) with a view to maintain the drug within the physique for longer, the opposite three domains map onto the three-part construction of the SARS-CoV-2 Spike protein. To be extra exact, the DARPins concurrently block the receptor binding area (RBD) of every spike trimer, permitting them to inhibit each tightly and cooperatively ACE2 interplay (FIGURE 1G). By blocking ACE2 interplay, ensovibep prevents SARS-CoV-2 from binding to our cells and, in so doing, protects them from an infection.
FIGURE 1. A) Schematic illustration of the five-part ensovibep assemble (half life extenders, H1 … [+]
Sylvia Rothenberger et al. 2021.
The three-part construction of ensovibep, mixed with the excessive binding affinity of DARPins, makes it troublesome for mutations to buck off. The one mutations that managed to take action, a substitution at F486V and one other at N234Q, come on the expense of viral health extra usually. For instance, with the F486V substitution, there was a ~8.5 fold discount in binding affinity between human ACE2 and the SARS-CoV-2 RBD. That form of discount would considerably cut back the virus’ means to contaminate our cells, lowering transmissibility and virulence. The significance of F486 is additional mirrored by a low frequency of naturally occurring substitutions.
Along with ensovibep’s potential to carry out towards variants, it has the advantage of being warmth steady —which means it may be saved and transported simply. Additionally, due to DARPins’ comparatively easy molecular structure, they are often manufactured in high yields, resulting in cost-effective and scalable manufacturing.
The primary disadvantage is that ensovibep needs to be delivered intravenously, proscribing its use to a medical setting and certain elevating its value. It additionally means it could’t be used prophylactically, to forestall an infection and to forestall transmission, which, realistically, is what we most want to manage this pandemic.
We’ll have to attend and see what image the outcomes from part 3 paint —which is able to embody a pattern dimension of 2100 sufferers and double down on testing the medical efficacy at 75mg— however up to now it’s trying like ensovibep will make a useful addition to our still-sparse quiver of antiviral medication.